By Evan C. Mascitti
Clay appears so simple. It’s dirt, dust and mud. It is the Earth itself. What could be more ordinary?
If we look closer, clay becomes complex and fascinating. Why does it form hardpan when dry but a soupy mess when wet? What makes some clays act so differently from others? To answer these, we first must wrestle with another riddle: what exactly is clay?
Faced with this question, many would offer something like the famous utterance by Justice Potter Stewart:
“I can’t define it, but I know it when I see it.”
This way of thinking about clay is perfectly suitable for practical, day-to-day workings with soil. For a more penetrating analysis, we must do a bit better.
What’s in a name?
How you define clay depends on who you are. Here is a long-winded, official statement maintained by the Clay Minerals Society:
“The term ‘clay’ refers to a naturally occurring material composed primarily of fine-grained minerals, which is generally plastic at appropriate water contents and will harden when dried or fired. Although clay usually contains phyllosilicates, it may contain other materials that impart plasticity and harden when dried or fired. Associated phases in clay may include materials that do not impart plasticity and organic matter.” (Guggenheim and Martin, 1995)
This is quite a mouthful, and it characterizes the banter typical among academic scientists. This kind of debate is hardly unique to soils. Biologists often have trouble defining life, and some astronomers disagree on what constitutes a planet. Detailed definitions could seem like useless squabbling, but there is a good reason for it: our human desire to put things into categories doesn’t always jive with nature’s complexity. We have to draw lines somewhere, and definitions ensure everyone is on the same page.
The long passage above captures three ways to think about the word “clay:” as a particle size, as a mineral, or as a behavior. Each is useful in its own way.
Clay as a particle size
Particle size is probably the most familiar definition of clay. Clay-size particles are 2 microns (0.002 mm) and smaller. Even this is not clear-cut – what is “size?” Does it mean height? Width? Volume? For example, which has the greater size: a Frisbee or an apple? The Frisbee is wider, but the apple is taller. Actually, I would argue that they are about the same size because their volumes are nearly equal. Only a perfect sphere can be described by a single “size” because all its dimensions are identical. Clay particles tend to be flat and wide (for reasons discussed below), so the “size” of a clay particle depends on how it is measured. To standardize the definition of size, soil scientists measure the settling of particles in a column of water. We assume the particles are spherical (even though they aren’t) and calculate their settling rates based on a mathematical principle called Stokes’ Law. Clay-size particles sink at the same rate as a sphere having a diameter of 2 microns. This is called the “equivalent spherical diameter,” or ESD.
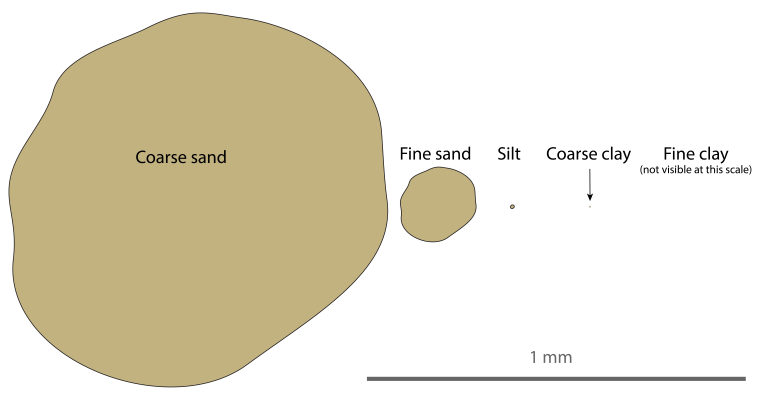
Clay particles are not just a little smaller than sand or silt. They are so much smaller that it is almost impossible to grasp. It takes serious mental effort visualize their size – even if you are a soil scientist who thinks about this every day. Figure 1 shows a scaled representation of some soil particles. The “coarse” clay is barely visible, and the “fine” clay (<0.2 microns ESD) cannot be drawn at this scale. The differences are magnified even further by comparing volume, which is the truest definition of “size.” By volume, a fine clay particle is 1 trillion times smaller than a sand grain. The word “trillion” is used casually these days by politicians and journalists, but I try hard to remember that one trillion is not just more than one billion, it is a thousand billion – a number that feels impossibly large.
Clay as a mineral
Soil particles are minerals. Minerals are made of atoms. This is the key to understanding the different types of clay.
Clay minerals are officially named phyllosilicates. Phyllo is a Greek root meaning “sheet-like,” and silicate refers to the element silicon. Clay minerals are sheet-like because their atomic bonds are stronger in the two horizontal directions than in the vertical direction (Figure 2). The particles tend to grow in the x and y directions and separate more easily in the z-dimension. Most clay mineral particles have an aspect ratio of at least 40:1, with some well over 100:1 (Dixon and Weed 1989). Figure 3 shows some platy kaolinite particles under an electron microscope.
The atomic arrangement also determines how a clay interacts with water. Some clay minerals have a net negative charge at their surfaces. These charged surfaces attract water molecules. High-charge clays like vermiculite and smectite hold water very tightly. Illite also has a high charge, but it has less surface area, so its overall affinity for water is intermediate. Kaolinite has no permanent charge.
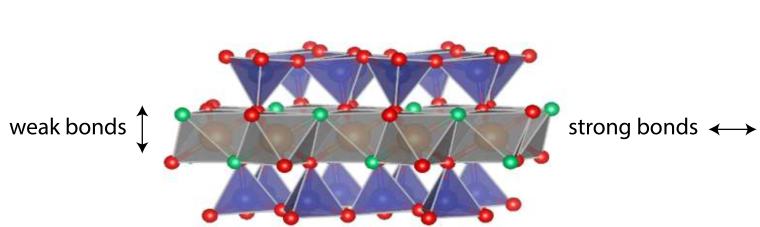
Figure 2: Clay minerals are strongly bonded in the x and y dimensions and weakly in the z-dimension, leading to their platy shape. Adapted from Wang et al. (2020).

Clay minerals usually occur as very small crystals, so the particle size and mineralogy definitions are easy to conflate. Soil scientists adopted the 2-micron silt/clay boundary in the 1930s because it is the smallest size, on average, at which non-clay minerals occur. There are still plenty of clay mineral particles larger than 2 microns, and plenty of non-clay minerals smaller than 2 microns. Before 1938, the clay size boundary was variously defined as 1, 5 or 15 microns (Simonson 1999). Therefore, the 2-micron cutoff is not as black and white as it is often considered.
Clay as a behavior
I believe thinking of clay as a behavior is the most useful definition for sports field managers. This definition is based on plasticity. Plasticity is the soil’s ability to be molded into a new shape. Plasticity is what allows potters to shape clay to their liking, and it is what allows a sports field manager to scratch and pack a pitcher’s mound.
In the behavioral definition of clay, particle size and mineralogy are ignored. They are the true drivers of soil behavior, but this information is not really needed to decide whether a soil “acts” more like a silt or a clay. Civil engineers use lab tests to quantitatively distinguish “clay-like” behavior from “silt-like” behavior. These tests measure the soil’s response when water is added or subtracted. There are two critical water contents: the plastic limit and the liquid limit. Figure 4 shows a sample at these thresholds.

At the plastic limit, the soil is just moist enough to be molded into a new shape. At the liquid limit, it begins to flow as a paste. The difference between the two is the plasticity index: it is the range of water contents over which the soil can be shaped and molded. Clays generally accept more water before becoming liquefied. Therefore, they have larger plasticity indices than silts.
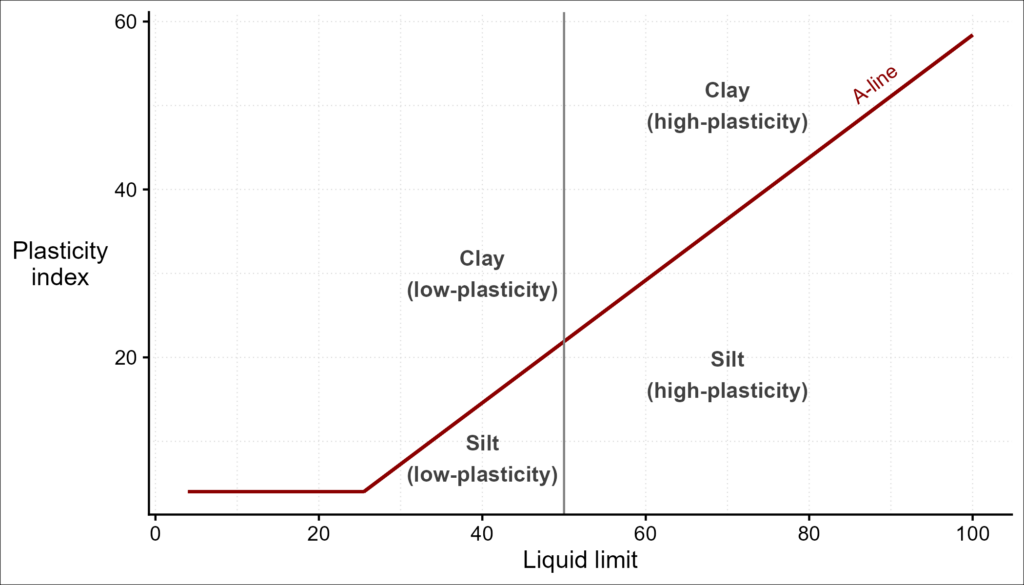
Figure 5 shows a plasticity chart. A given soil is considered clay if it plots above the A-line. It is classified as a silt if it plots below the A-line. Both clays and silts can be further described as having high or low plasticity. The position of a soil on this chart helps predict its physical properties such as drainage and stiffness. The “clayeyness” of a soil increases from lower left to upper right.
Prof. Arthur Casagrande developed this system during World War II. In 1942 the United States was desperate to build air bases in the Pacific isles. Casagrande’s system helped military engineers ensure the soil below their runways would support heavy bombers during takeoff and landing. The tests required little equipment, which meant they could be performed in makeshift laboratories right on the island. More importantly, the tests directly recorded how the soil behaved when it got wet.
Soils that behave as clays are excellent for skin areas, but they are not conducive to growing healthy grass. Clays impede drainage and root respiration when compacted. When dry, their high strength prevents roots from penetrating. Clays retain more moisture than silts, but much of the water is held too tightly for plants to use.
What is clay? There is no single answer.
There are three ways to think about clay: as a particle size, as a family of minerals, and as a behavior. The first two are most useful to scientists, and the latter might better relate to sports surface performance. All three are important because the particle size and mineralogy are what ultimately dictate the soil behavior we observe and depend on.
Thinking on the micro scale is hard, but worthwhile. The performance of clay goes all the way back to atoms. That idea is both useful and fun!
Evan C. Mascitti is a Ph.D. candidate in soil science at Penn State University. He previously worked for several years as a grounds manager in professional baseball.